The brain changes associated with Alzheimer’s usually lead to a severe loss of memory and cognitive abilities, but not always.
Now, a new study led by a team at the University of California, San Diego (UCSD) helps explain why.
Based on postmortem brain research, around 20 to 30 percent of older adults are thought to have asymptomatic Alzheimer’s disease (ASYMAD). Their brains have the characteristic buildup of misfolded amyloid-beta and tau proteins, but they show no detrimental mental effects.
“Even when the brain shows clear signs of Alzheimer’s, some people stay mentally sharp,” says UCSD medical scientist Sushil Mahata.
In their new study in mice, Mahata and colleagues have discovered a molecular switch that may be responsible for connecting these amyloid-beta plaques and tau tangles to cognitive decline.
While the findings need to be further confirmed in human patients with Alzheimer’s, it’s a significant advance in our understanding of ASYMAD, and could aid the development of treatments to tackle Alzheimer’s symptoms.
“This work establishes a mechanistically grounded framework for studying cognitive resilience in Alzheimer’s disease and provides a scalable platform for interrogating sex-specific protective pathways, identifying early biomarkers of disease trajectory, and enabling mechanism-guided development of preventive therapeutic strategies,” write the researchers in their published paper.
To begin with, the researchers deployed an AI-powered scanning technique on genetic data points covering thousands of postmortem human brain samples – from people who died both with and without Alzheimer’s disease.
That gave the team a bank of around 40 different genes that together formed a ‘fingerprint’ of Alzheimer’s. They used this fingerprint to assess both healthy lab mice and those bred to develop Alzheimer’s-like brain pathology.
The mouse tests confirmed the fingerprint could distinguish between these groups, but they also revealed something else.
Using an AI-based model, the researchers identified the protein chromogranin A (CgA) as a key driver in this gene network. When the researchers bred mice lacking the CgA protein, the animals still developed the biological hallmarks of Alzheimer’s without the symptoms.
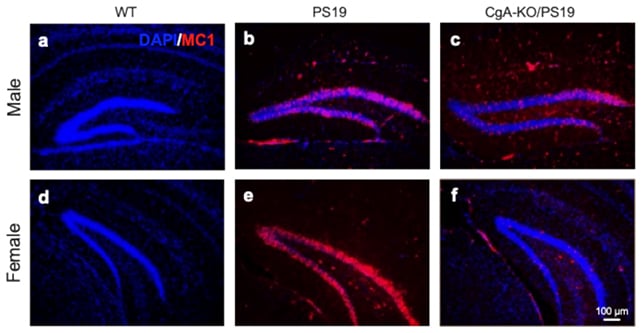
CgA is involved in brain cell communication, but it has also been suspected of influencing Alzheimer’s progression. These new discoveries suggest it may actually be crucial in asymptomatic Alzheimer’s.
There was a notable difference between the sexes too. While male mice without CgA matched the genetic patterns of Alzheimer’s disease without any memory or learning problems, female mice without CgA showed even fewer signs of Alzheimer’s-related brain damage.
The reason for that difference isn’t clear.
The researchers suggest it may be related to sex differences in hormones, immune systems, or genetics, and it’s an area for future studies to follow up on.

“Collectively, these findings support a paradigm in which understanding and harnessing endogenous resilience mechanisms may represent a critical path forward for altering the course of Alzheimer’s disease,” writes the team.
Exactly why CgA is so important remains to be determined. Based on their previous work, the researchers think it might act as a “molecular amplifier” of toxic proteins in the brain, and that removing or limiting it could protect against cognitive decline.
It’s worth noting that, in addition to having spent several years analyzing its biological effects, some of the study authors are invested in a company that is pioneering research into CgA in relation to Alzheimer’s.
Moving forward, any potential treatments involving regulating CgA are still a long way off – and they would need to ensure that all the normal, healthy functions the protein carries out can continue as normal.
However, we now have solid evidence that CgA is significant in linking the abnormal buildup of amyloid-beta and tau proteins to Alzheimer’s symptoms – and we also have a mouse model ready for further study of the condition.
Related: Blood Tests Could Reveal Alzheimer’s Risk Years Before Symptoms Appear
“We’re beginning to uncover the brain’s built-in defenses,” says Mahata, who is listed as a founder of CgA Therapeuticals.
“And that could fundamentally change how we approach treatment.”
The research has been published in Acta Neuropathologica Communications.